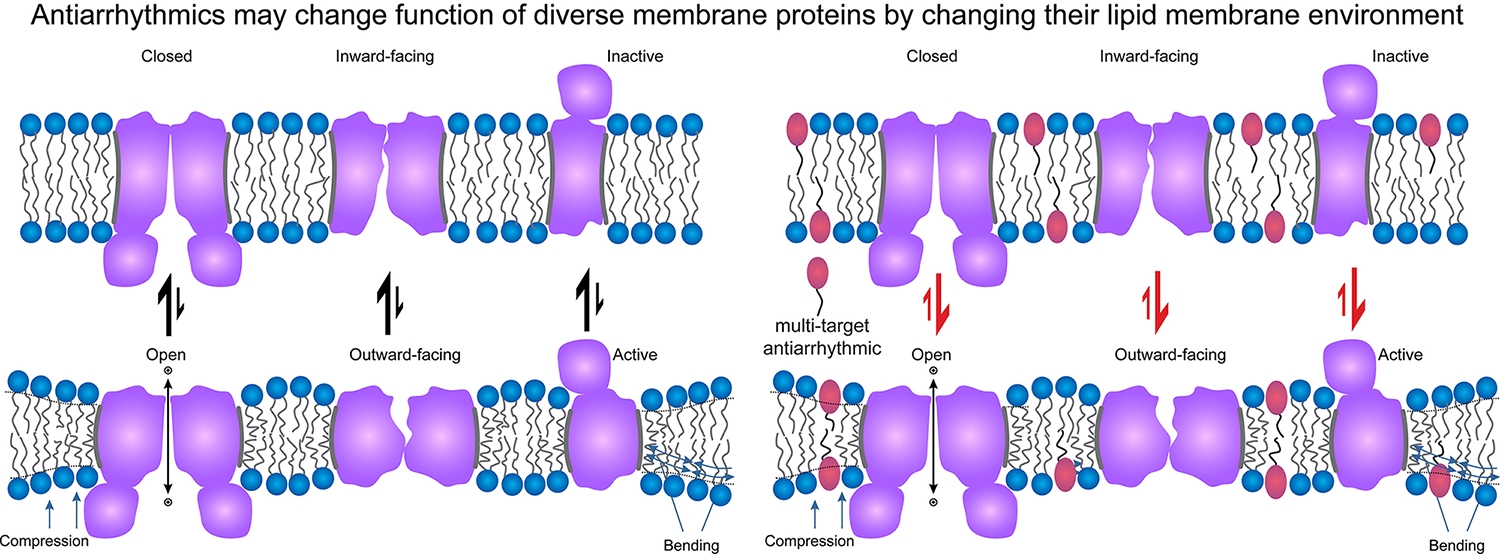
Clinicians have long observed that using a "dirty drug" that hits a number of targets instead of just one is the most efficacious approach to treat conditions that cause abnormal heart rhythms. Now, Weill Cornell Medicine researchers have discovered how this kind of drug becomes "dirty."
In the study, published Nov. 16 in The Journal of General Physiology, the investigators identified a possible mechanism involved in the beneficial, as well as toxic, effects of the most popular of the multi-target, antiarrhythmic drugs, amiodarone. These findings may not only impact what drugs doctors prescribe to the approximately 2.7 million American patients with atrial fibrillation — a condition characterized by a quivering, irregular heartbeat that's a major risk factor for stroke — but also lead to more research on this type of drug to guide the development of better options with fewer side effects.
"If you were in the business of bringing new drugs to market, a drug like amiodarone, which takes this promiscuous approach, looks pretty scary," said senior author Dr. Olaf S. Andersen, a Professor of Physiology and Biophysics at Weill Cornell Medicine and director of the Tri-Institutional M.D.-Ph.D. Program. "But when you have a system that is as complicated as the one that regulates rhythm in the heart, it may actually be advantageous to have a number of different targets."
"Until now, there has really been no understanding of how amiodarone can alter so many targets, and why this is important," said first author Dr. Radda Rusinova, an instructor in physiology and biophysics at Weill Cornell Medicine. "Now we have a solid hypothesis that can be followed up on in future studies, and a starting point to figure out how to change this drug to make it better at doing its job."
The investigators, including a collaborator from the University of Arkansas, studied four antiarrhythmic drugs — amiodarone, dronedarone, propranolol and pindolol. Amiodarone was developed in 1967 and is still considered the most effective of the lot, but little was known about how it simultaneously regulates a number of membrane proteins, including potassium channels, other ion channels, and hormone receptors. If one could determine how and why amiodarone works, other scientists down the line could alter it, with the goal of achieving the same outcomes with fewer serious side effects, like hyperthyroidism and negative impacts on the liver, lungs and eyes.
To conduct the work, they used simple synthetic lipid environments, as well as mixtures of lipids to create an environment that's more cell-like — and therefore human-like. They found that at doses that typically would be used therapeutically, amiodarone affects the lipid bilayer — an outside envelope of the cell where membrane proteins reside, which in turn can affect the proteins themselves.
Like amiodarone, dronedarone — which was developed to have fewer side effects — also altered lipid bilayer properties; it was in fact more potent than amiodarone, which might cause dronedarone to become toxic at lower concentrations than amiodarone. This may explain why dronedarone is considered less effective as an antiarrhythmic. Despite seemingly good effects in cellular electrophysiological studies, it becomes difficult to administer safely because it is toxic at lower concentrations, which limits the dosage such that it may take too long to reach effective levels.
"Some drugs just have the right effects on a number of different targets. Amiodarone is one of those drugs," Dr. Andersen said. "That's our key takeaway: Dirty drugs may be good for you."